Have the “Protein Police” been dethroned by the Ace of Amino Acids, BCAA’s?
The functioning and cultivating of skeletal muscle lies at the core of our profession. As trainers, we tend to focus on aesthetics and health when working with clients, since these seem to be among the most prevalent reasons individuals give for coming to the gym.
However, we would be remiss in our duties as knowledgeable practitioners if we oversimplify the role of skeletal muscle by describing it merely as the force behind the body’s ability to move.
Skeletal muscle is also the main holding tank of amino acids which are utilized by other bodily tissues. These amino acid reserves are the only resources in the human body that can withstand significant depletion without compromising the ability to sustain life.
The Exercise-Amino Acid Connection
When the body experiences a drop in blood sugar, amino acids come to the rescue as the liver’s primary substrate in manufacturing additional glycogen stores. Specifically, it is the nitrogen contained within amino acids that is a key component of DNA and the myriad of other molecules necessary for cell maintenance and replication.
Tissue demands for amino acids skyrocket when the body is under stress, which can be categorized as severe illness, traumatic injury or, most applicable to our profession, a serious strength-training regimen. Intense physical activity and recovery are contributors to the significant changes observed in amino acid and protein metabolism within skeletal muscle. It is during these times that the body has a particular demand for branched-chain amino acids, often referenced as BCAAs.
During exercise, muscle protein synthesis decreases as protein degradation increases. As activity levels continue to rise, there is a reduction in the concentration of branched-chain amino acids circulating in the blood plasma as well as within the cells themselves. After a tough training session, therefore, recovery of muscle protein synthesis requires a rescue supply of dietary protein or BCAAs to restore these levels to their former homeostatic balance.
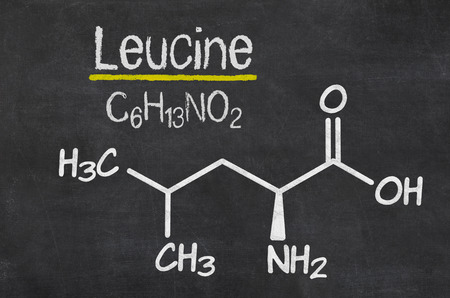
Leucine’s Leading Role
BCAA’s are unique in that the body cannot manufacture them on its own, and must be obtained from either dietary sources or direct supplementation. In the past, researchers viewed amino acids simply as substances required for the manufacture of muscle and other body proteins—similar to the bricks required to build a house.
Most studies refer specifically to the BCAAs leucine, isoleucine, and valine. Since these are the only three amino acids capable of being utilized as “fuel” by muscle tissues, both blood and muscle levels of BCAA’s suffer a significant depletion by the end of an intense exercise session. In the case of leucine, however, recent investigations have proven this amino acid to play multiple roles that go well beyond simply providing material to build muscle.
Of the three aforementioned BCAAs, leucine is by far the most abundant, having a 10-fold greater impact on protein synthesis than any other amino acid.
Solo Act?
BCAAs are preferentially burned as energy by muscles to fuel work output. It is now widely accepted that whey protein is a very rich source of BCAAs, explaining its popularity as a main ingredient in many post-workout supplement shakes. Researchers who study why protein consumption helps stimulate anabolism (the building of muscle) have been able to demonstrate that it is the level of amino acids in the bloodstream that has the greatest impact on boosting protein synthesis in muscles. In particular, the levels of the BCAA leucine seem to correlate the most with protein synthesis.
In a series of brilliantly controlled and executed experiments, researchers examined levels of muscle protein synthesis after subjects’ consumption of various formulations of amino acids, and compared them to control results observed after the ingestion of glucose alone. When a complete protein (one that contains all the amino acids) was consumed, protein synthesis increased as expected.
Consuming only essential amino acids netted the same increase, suggesting that non-essential amino acids may not be specifically required to stimulate an anabolic state. When BCAAs were consumed in the absence of any other form of supplementation, the same anabolic spike was once again observed.
The final experiment in the series, the crowning glory, consisted of having the subjects refuel with leucine alone. Remarkably, protein synthesis still increased on the same order of magnitude! These findings provided strong evidence that leucine is the driving force behind the ability of dietary protein to stimulate protein synthesis.
There is an interesting possible explanation for these results. Whole proteins take a fairly long time to empty from the stomach into the small intestine and ultimately into circulation, where they can be utilized effectively. Even a fast-digesting protein such as whey can take hours to liberate the leucine therein, allowing it to enter circulation.
An isolated free-form leucine supplement, however, becomes quickly absorbed into the bloodstream, thus spiking plasma leucine levels and drastically increasing intracellular leucine concentrations. We have learned that the quality of a protein source is primarily dependent upon its leucine content; therefore, for the body to re-set itself into the desired positive nitrogen balance after a workout, a source of leucine must be consumed.
In an environment devoid of sufficient amounts of this BCAA, protein balance will remain negative, causing the process of muscle anabolism to suffer. This lends credence to the data showing how ingesting BCAAs prior to engaging in resistance exercise can reduce muscle soreness.
Bodies Brilliantly Balance BCAA’s
One of the most researched pathways of muscle growth is called the mTOR pathway (mechanistic target of rapamycin). This process is highly dependent upon and sensitive to circulating concentrations of leucine. When low levels of this BCAA are detected, the body signals mTOR that the environment is experiencing a lack of dietary protein, and as such cannot synthesize new skeletal muscle. When this occurs, the body simply disables the mTOR pathway.
Once sufficient concentrations of leucine are consumed, mTOR receives the signal that adequate dietary protein is now present and switches on overall protein synthesis. Since an increase in mTOR activity results in more muscular growth, it is easy to see how important leucine is in the overall scheme of a positive strength-training outcome.
Many trainers are not comfortable recommending workout supplementation to their clients, realizing prudently that this may extend beyond their professional scope of practice. However, if the occasion should arise when a client is trying to decide between 2 different supplements, and upon reading the ingredients of each it becomes clear that you can point him in the proper direction, take the time to educate him. The client will benefit and you can feel good about helping him on his healthy journey.
References:
- https://www.runtastic.com/blog/en/nutrition/do-runners-need-dairy-to-lose-weight/
- http://jn.nutrition.org/content/136/2/533S.short
- https://nutritionandmetabolism.biomedcentral.com/articles/10.1186/1743-7075-9-67
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2732256/
- https://www.bodybuilding.com/fun/supplemental-leucine-how-it-powers-muscle-growth.html
- https://www.just-fly-sports.com/role-of-leucine/
- https://www.bodybuilding.com/fun/layne39.htm
- http://www.fitness1sttraining.com/single-post/2014/08/01/Protein-Synthesis